|
The difference between energies for a cutoff energy of 775 eV and 900 eV is roughly 0.00057 eV. 2 shows that a cutoff energy of 775 eV is good enough to obtain a roughly constant energy. 2,3,4,5 the energy values on y axis are relative to the lowest energy obtained during cutoff energy optimization.Ĭut-off energy was then optimized for 12x12x12 grid on a BCC lattice with 346 pm as the lattice constant. While optimizing number of k points, the cut-off energy was held constant at the default value of 408 eV. 1 are the differences between energies of the current no. 1 shows that a 12x12x12 grid in k-space is sufficient for an accuracy of 0.005 eV. Moreover, lowest lattice constant requires the highest number of k points and hence 346 pm was chosen for optimizing the number of k points. Since 351 pm is the lattice constant of BCC at room temperature, varying the lattice constant from 346 pm to 356 pm would be sufficient to get an optimum lattice constant at which the energy is minimized. BCC lattice was chosen because at room temperature lithium crystallizes as BCC with a lattice constant of 351 pm. The number of k points was optimized for BCC lattice with a lattice constant of 346 pm. However, before varying the lattice parameter for a given crystal structure, the optimum number of k points and the cutoff energy to be used in calculations were obtained. Minimum energy of different crystal structures was compared to obtain the optimum crystal structure and the corresponding lattice parameter. A core radius of 1 Bohr and an electronic configuration of 1s2 2s1 was used for calculations. For all calculations Generalized Gradient Approximation (GGA) and Perdew-Burke-Ernzerhof (PBE) exchange correlation functionals were used with On The Fly Generated (OTFG) ultrasoft pseudopotential. In order to study the crystal structure of lithium, energy was minimized for different crystal structures by using CASTEP in Material studio. Moreover, repeating calculations in a software is also much cheaper than repeating an experiment. Density Functional Theory (DFT) was used for studying the crystal structure as it is much cheaper than using experimental techniques such as X-ray diffraction. Hence, lithium is one of the most important metals and so the crystal structure of lithium was studied in this project. and lithium metal batteries are amongst the most promising future energy storage technologies. 
Today, lithium ion batteries are being used in variety of applications such as electric vehicles, laptops, mobile phones, grid storage etc. Lithium is a very important component of batteries today. However, SCC has a significantly higher energy compared to BCC and FCC. A comparison of 0 K lattice energies at the optimum lattice parameter suggests that the energy difference between BCC and FCC is lower than 0.005 eV, with BCC having lower energy. Optimum lattice parameters have been obtained for BCC, SCC, and FCC lattices by using plane-wave basis set density functional theory methods. The polarizability of the anion will be high if the size is large and the charge is maximum.In this project, ‘Material Studio’ has been used to study the crystal structure of lithium by using Density Functional Theory (DFT). The polarizing power of the cation is high if the size is small and the charge is maximum. According to Fajan’s rule, the covalent character in ionic compounds are directly proportional to the polarizing power of the cation and inversely proportional to the polarizability of the anion. 
The covalent character in ionic compounds is determined by using Fajan’s rule. Thermodynamic stability is inversely proportional to the covalent character. Note: Thermodynamic stability is directly proportional to the ionic character. Lattice energy $ \propto $ Charge of the metal On increasing the size of the negative ion the lattice energy decreases. On increasing the charge of the metal lattice energy increases. Lattice energy depends upon the charge and size. Thermodynamics stability $ \propto $ Lattice energy. On increasing the lattice energy thermodynamics stability increases. The thermodynamics stability of the oxides and carbonated depends upon the lattice energy. 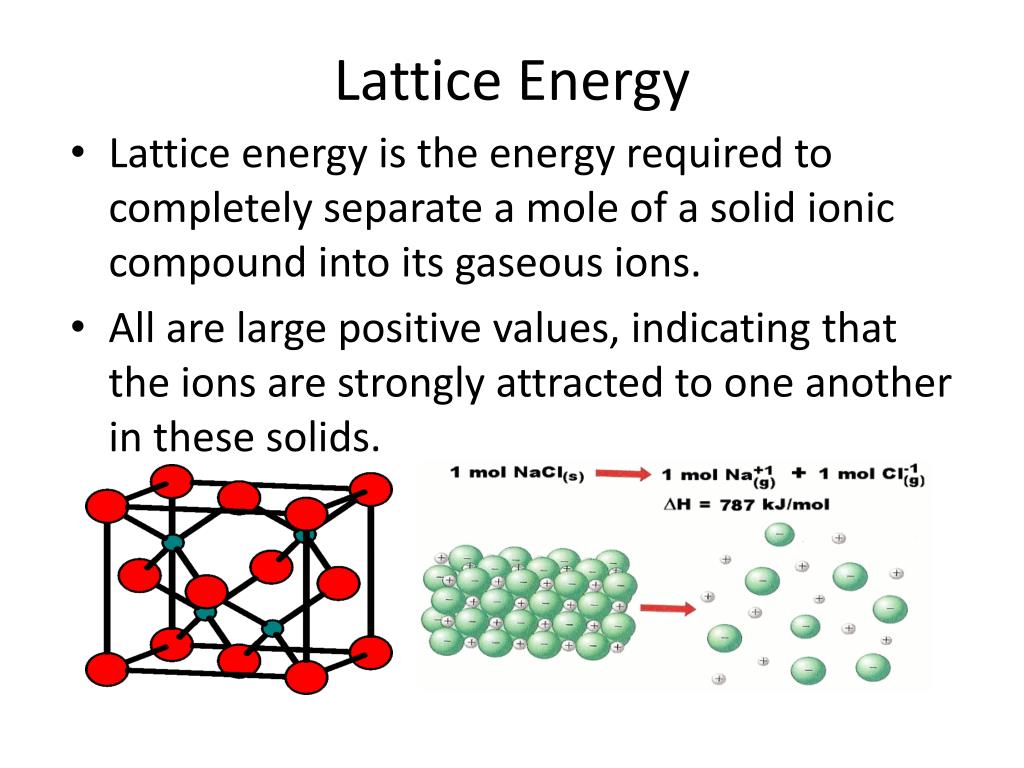
Lattice energy is directly proportional to the charge and inversely proportional to the size. 
Thermodynamics stability is directly proportional to the lattice energy. Hint: We will determine the thermodynamic stability of the lithium oxide and lithium carbonate.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
